
KEYTRUDA® is indicated in combination with pemetrexed and platinum chemotherapy for the first-line treatment of patients with metastatic non-squamous non-small cell lung cancer (NSCLC), with no EGFR or ALK genomic tumor aberrations. In combination with carboplatin and either paclitaxel or paclitaxel protein-bound indicated for the first-line treatment of patients with metastatic squamous NSCLC. Single agent indicated for the first-line treatment of patients with NSCLC expressing PD-L1 [Tumor Proportion Score (TPS) ≥1%] as determined by a validated test, with no EGFR or ALK genomic tumor aberrations, and is either stage III where patients are not candidates for surgical resection or definitive chemoradiation, or metastatic. Single agent indicated for the treatment of patients with mNSCLC whose tumors express PD-L1 (TPS ≥1%) as determined by a validated test, with disease progression on or after platinum-containing chemotherapy (patients with EGFR or ALK genomic tumor aberrations should have disease progression on FDA-approved therapy for these aberrations prior to receiving KEYTRUDA®). Single agent indicated for adjuvant treatment following resection and platinum-based chemotherapy for adult patients with stage IB (T2a ≥4 cm), II, or IIIA NSCLC. Treatment of patients with resectable (tumors ≥4 cm or node positive) NSCLC in combination with platinum-containing chemotherapy as neoadjuvant treatment, and then continued as a single agent as adjuvant treatment after surgery.1

The durable benefits of KEYTRUDA® further recognized by ESMO

The latest 2024 ESMO MCBS scorecard update acknowledges the potential for curability with KEYTRUDA®.2
KEYTRUDA® has made significant advancements on the treatment of mNSCLC to receive an ‘A’ score on the ESMO MCBS, which is the highest grade in the curative setting.2,3


What do the ESMO MCBS scorecards tell us?3
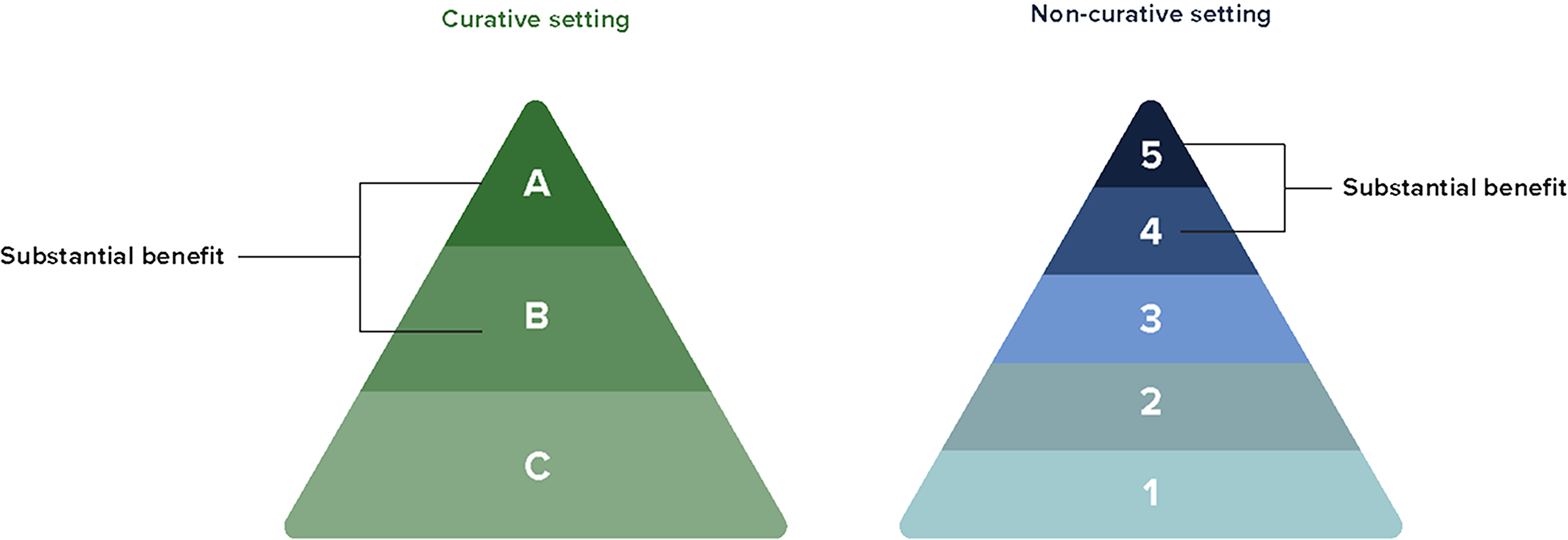
To learn more about the use of these scorecards in clinical practice, please click here.
1L: first-line; ALK: anaplastic lymphoma kinase; EGFR: epidermal growth factor receptor; ESMO: European Society for Medical Oncology; FDA: United States Food and Drug Administration; KN: keynote; MCBS: Magnitude of Clinical Benefit Scale; mNSCLC: metastatic non-small cell lung cancer; NSCLC: non-small cell lung cancer; PD-L1: programmed death ligand 1; TPS: tumor proportion score.

We keep moving forward, so that progress for our patients never stops

KEYTRUDA® changed the treatment landscape of NSCLC, offering more hope to your eligible patients for more time spent with loved ones,4-6 without impacting quality of life1,7-10
Watch how KEYTRUDA® has helped more patients

NSCLC: non-small cell lung cancer.

Gaining evidence and experience since 2011
Since the beginning of the phase I trial in 2011, KEYTRUDA® has received a growing number of approvals backed by robust clinical trial data, to reach more of your patients.1,11

Reshaping NSCLC treatment

KEYTRUDA® was the FDA-approved immune checkpoint inhibitor (ICI) for 1L treatment of lung cancer12
With evidence and experience gained in mNSCLC since 2011, the impact of KEYTRUDA® is evident.11
KEYTRUDA® is FDA-approved ICI for lung cancer and has recently achieved a 5-year OS milestone in mNSCLC.4-6,12
For use in both squamous and non-squamous mNSCLC, KEYTRUDA® is recognized by international guidelines:13-15
NCCN® 2025

One of the preferred 1L treatment options* (Category 1)
ASCO 2025

Strongly recommended 1L treatment option
(Strong)
ESMO 2023

One of the standard 1L treatment options
(I,A)
*NCCN® makes no warranties of any kind whatsoever regarding their content, use or application and disclaims any responsibility for their application or use in any way.15
1L: first-line; ASCO: American Society of Clinical Oncology; ESMO: European Society for Medical Oncology; FDA: United States Food and Drug Administration; ICI: immune checkpoint inhibitor; KN: keynote; mNSCLC: metastatic non-small cell lung cancer; NCCN®: National Comprehensive Cancer Network®; NSCLC: non-small cell lung cancer; OS: overall survival.

At MSD, our patients are our compass
MSD cares about helping people live better lives, offering a durable treatment without compromising quality of life for mNSCLC patients, with KEYTRUDA®.1,4-10,16
The recent expansion of indications means that KEYTRUDA® may offer you a treatment option for a broader range of eligible patients:1
across advanced or earlier stages of NSCLC
across histologies (NSq/Sq)
as monotherapy or in combination with chemotherapy
with Q3W or Q6W dosing options
mNSCLC: metastatic non-small cell lung cancer; NSCLC: non-small cell lung cancer; NSq: non-squamous; Q3W: every three weeks; Q6W: every six weeks; Sq: squamous.

Continuing to help patients around the world
The milestones KEYTRUDA® has achieved are a testament to our commitment to offering treatment possibilities for more people living with NSCLC.1,17-19

Approval for use in
More than 95 countries

6 Egypt-approved
NSCLC indications

6 FDA-approved
NSCLC indications

6 EMA-approved
indications for NSCLC
EMA: European Medicines Agency; FDA: United States Food and Drug Administration; NSCLC: non-small cell lung cancer; UAE: United Arab Emirates.
KEYTRUDA® (pembrolizumab) injection, for intravenous use
KEYTRUDA® is a programmed death receptor-1 (PD-1)-blocking antibody indicated:
Melanoma
- For the treatment of patients with unresectable or metastatic melanoma.
- For the adjuvant treatment of adult and pediatric (12 years and older) patients with Stage IIB, IIC, or Ill melanoma following complete resection.
Non-Small Cell Lung Cancer (NSCLC)
- In combination with pemetrexed and platinum chemotherapy, as first-line treatment of patients with metastatic nonsquamous NSCLC, with no EGFR or ALK genomic tumor aberrations.
- In combination with carboplatin and either paclitaxel or paclitaxel protein-bound, as first-line treatment of patients with metastatic squamous NSCLC.
-
As a single agent for the first-line treatment of patients with NSCLC expressing PD-L1 [Tumor Proportion Score (TPS) ≥ 1%] as determined by a validated test, with no EGFR or ALK genomic tumor aberrations, and is:
- Stage Ill where patients are not candidates for surgical resection or definitive chemoradiation, or
- a metastatic.
- As a single agent for the treatment of patients with metastatic NSCLC whose tumors express PD-L1 (TPS ≥ 1 %) as determined by a validated test, with disease progression on or after platinum-containing chemotherapy. Patients with EGFR or ALK genomic tumor aberrations should have disease progression on FDA-approved therapy for these aberrations prior to receiving KEYTRUDA.
- For the treatment of patients with resectable (tumors≥ 4 cm or node positive) NSCLC in combination with platinum-containing chemotherapy as neoadjuvant treatment, and then continued as a single agent as adjuvant treatment after surgery.
- As a single agent, for adjuvant treatment following resection and platinum-based chemotherapy for adult patients with Stage IB (T2a ≥ 4 cm), II, or IIIA NSCLC.
Malignant Pleural Mesothelioma (MPM)
- In combination with pemetrexed and platinum chemotherapy, as first-line treatment of adult patients with unresectable advanced or metastatic MPM.
Head and Neck Squamous Cell Cancer (HNSCC)
- In combination with platinum and FU for the first-line treatment of patients with metastatic or with unresectable, recurrent HNSCC.
- As a single agent for the first-line treatment of patients with metastatic or with unresectable, recurrent HNSCC whose tumors express PD-L1 [Combined Positive Score (CPS ≥ 1] as determined by a validated test.
- As a single agent for the treatment of patients with recurrent or metastatic HNSCC with disease progression on or after platinum-containing chemotherapy.
Classical Hodgkin Lymphoma (CHL)
- For the treatment of adult patients with relapsed or refractory cHL.
- For the treatment of pediatric patients with refractory cHL, or cHL that has relapsed after 2 or more lines of therapy.
Urothelial Cancer
- In combination with enfortumab vedotin, for the treatment of adult patients with locally advanced or metastatic urothelial cancer.
-
As a single agent for the treatment of patients with locally advanced or metastatic urothelial carcinoma who:
- are not eligible for any platinum-containing chemotherapy, or
- have disease progression during or following platinum-containing chemotherapy or within 12 months of neoadjuvant or adjuvant treatment with platinum-containing chemotherapy.
Microsatellite Instability-High or Mismatch Repair Deficient Cancer
- For the treatment of adult and pediatric patients with unresectable or metastatic microsatellite instability-high (MSI-H) or mismatch repair deficient (dMMR) solid tumors, as determined by a validated test, that have progressed following prior treatment and who have no satisfactory alternative treatment options.
Microsatellite Instability-High or Mismatch Repair Deficient Colorectal Cancer (CRC)
- For the treatment of patients with unresectable or metastatic MSI-H or dMMR colorectal cancer (CRC) as determined by a validated test.
Gastric Cancer
- In combination with trastuzumab, fluoropyrimidine- and platinum-containing chemotherapy, for the first-line treatment of adults with locally advanced unresectable or metastatic HER2-positive gastric or gastroesophageal junction (GEJ) adenocarcinoma whose tumors express PD-L1 (CPS≥ 1) as determined by a validated test.1
- In combination with fluoropyrimidine- and platinum-containing chemotherapy, for the first-line treatment of adults with locally advanced unresectable or metastatic HER2-negative gastric or gastroesophageal junction (GEJ) adenocarcinoma.
Esophageal Cancer
- KEYTRUDA, in combination with platinum- and fluoropyrimidine-based chemotherapy is indicated for the treatment of patients with locally advanced or metastatic esophageal or gastroesophageal junction (GEJ) (tumors with epicenter 1 to 5 centimeters above the GEJ) carcinoma that is not amenable to surgical resection or definitive chemoradiation.
Cervical Cancer
- In combination with chemoradiotherapy, for the treatment of patients with FIGO 2014 Stage III-IVA cervical cancer.
- In combination with chemotherapy, with or without bevacizumab, for the treatment of patients with persistent, recurrent, or metastatic cervical cancer whose tumors express PD-L1 (CPS ≥1) as determined by a validated test.
Hepatocellular Carcinoma (HCC)
- For the treatment of patients with HCC secondary to hepatitis B who have received prior systemic therapy other than a PD-1/PD-L1-containing regimen.
Biliary Tract Cancer (BTC)
- In combination with gemcitabine and cisplatin, for the treatment of patients with locally advanced unresectable or metastatic biliary tract cancer.
Merkel Cell Carcinoma (MCC)
- For the treatment of adult and pediatric patients with recurrent locally advanced or metastatic Merkel cell carcinoma.
Renal Cell Carcinoma (RCC)
- In combination with axitinib, for the first-line treatment of adult patients with advanced RCC.
- In combination with lenvatinib, for the first-line treatment of adult patients with advanced RCC.
- For the adjuvant treatment of patients with RCC at intermediate-high or high risk of recurrence following nephrectomy, or following nephrectomy and resection of metastatic lesions.
Endometrial Carcinoma
- In combination with carboplatin and paclitaxel, followed by KEYTRUDA as a single agent, for the treatment of adult patients with primary advanced or recurrent endometrial carcinoma.
- In combination with lenvatinib, for the treatment of adult patients with advanced endometrial carcinoma that is mismatch repair proficient (pMMR) as determined by validated test or not MSI-H, who have disease progression following prior systemic therapy in any setting and are not candidates for curative surgery or radiation.
Triple-Negative Breast Cancer (TNBC)
- For the treatment of patients with high-risk early-stage TNBC in combination with chemotherapy as neoadjuvant treatment, and then continued as a single agent as adjuvant treatment after surgery.
- In combination with chemotherapy, for the treatment of patients with locally recurrent unresectable or metastatic TNBC whose tumors express PD-L1 (CPS ≥ 10) as determined by a validated test.
Adult Classical Hodgkin Lymphoma: Additional Dosing Regimen of 400 mg Every 6 Weeks
- For use at an additional recommended dosage of 400 mg every 6 weeks for Classical Hodgkin Lymphoma.2
1This indication is approved under accelerated approval based on tumor response rate and durability of response. Continued approval for this indication may be contingent upon verification and description of clinical benefit in the confirmatory trials.
2This indication is approved under accelerated approval based on pharmacokinetic data, the relationship of exposure to efficacy, and the relationship of exposure to safety. Continued approval for this dosing may be contingent upon verification and description of clinical benefit in the confirmatory trials.
DOSAGE FORMS AND STRENGTHS
- Injection: 100 mg/4 ml (25 mg/ml) solution in a single-dose vial.
CONTRAINDICATIONS
- None.
WARNINGS AND PRECAUTIONS
-
Immune-Mediated Adverse Reactions
- Immune-mediated adverse reactions, which may be severe or fatal, can occur in any organ system or tissue, including the following: immune-mediated pneumonitis, immune-mediated colitis, immune-mediated hepatitis, immune-mediated endocrinopathies, immune-mediated nephritis with renal dysfunction, immune-mediated dermatologic adverse reactions, and solid organ transplant rejection.
- Monitor for early identification and management. Evaluate liver enzymes, creatinine, and thyroid function at baseline and periodically during treatment.
- Withhold or permanently discontinue based on severity and type of reaction. Infusion-related reactions: Interrupt, slow the rate of infusion, or permanently discontinue KEYTRUDA based on the severity of reaction.
- Complications of allogeneic HSCT: Fatal and other serious complications can occur in patients who receive allogeneic HSCT before or after being treated with a PD-1/PD-L1 blocking antibody.
- Treatment of patients with multiple myeloma with a PD-1 or PD-L1 blocking antibody combination with a thalidomide analogue plus dexamethasone is not recommended outside of controlled clinical trials.
- Embryo-Fetal toxicity: Can cause fetal harm. Advise females of reproductive potential of the potential risk to a fetus and to use effective method of contraception.
ADVERSE REACTIONS
Most common adverse reactions (reported in ≥ 20% of patients) were:
- KEYTRUDA as a single agent: fatigue, musculoskeletal pain, rash, diarrhea, pyrexia, cough, decreased appetite, pruritus, dyspnea, constipation, nausea, and hypothyroidism.
- KEYTRUDA in combination with chemotherapy or chemoradiotherapy: fatigue/asthenia, nausea, constipation, diarrhea, decreased appetite, rash, vomiting, cough, dyspnea, pyrexia, alopecia, peripheral neuropathy, mucosal inflammation, stomatitis, headache, weight loss, abdominal pain, arthralgia, myalgia, insomnia, palmar-plantar erythrodysesthesia, urinary tract infection, and hypothyroidism.
- KEYTRUDA in combination with chemotherapy and bevacizumab: peripheral neuropathy, alopecia, anemia, fatigue/asthenia, nausea, neutropenia, diarrhea, hypertension, thrombocytopenia, constipation, arthralgia, vomiting, urinary tract infection, rash, leukopenia, hypothyroidism, and decreased appetite.
- KEYTRUDA in combination with axitinib: diarrhea, fatigue/asthenia, hypertension, hepatotoxicity, hypothyroidism, decreased appetite, palmar-plantar erythrodysesthesia, nausea, stomatitis/mucosal inflammation, dysphonia, rash, cough, and constipation.
- KEYTRUDA in combination with lenvatinib: hypothyroidism, hypertension, fatigue, diarrhea, musculoskeletal disorders, nausea, decreased appetite, vomiting, stomatitis, weight loss, abdominal pain, urinary tract infection, proteinuria, constipation, headache, hemorrhagic events, palmar-plantar erythrodysesthesia, dysphonia, rash, hepatotoxicity, and acute kidney injury.
- KEYTRUDA in combination with enfortumab vedotin: rash, peripheral neuropathy, fatigue, pruritus, diarrhea, alopecia, weight loss, decreased appetite, dry eye, nausea, constipation, dysgeusia, and urinary tract infection.
USE IN SPECIFIC POPULATIONS
Lactation:
- Advise women not to breastfeed during treatment with KEYTRUDA® and for 4 months after the final dose.
Pregnancy:
- Pembrolizumab has the potential to be transmitted from the mother to the developing fetus. Advise pregnant women of the potential risk to a fetus.
Fertility:
- Fertility studies have not been conducted with pembrolizumab.
- Egyptian Drug Authority KEYTRUDA® leaflet approval date 30/12/2024.
- Always read the full prescribing information.
- Healthcare professionals are asked to report any suspected adverse reactions to Egyptian Pharmacovigilance Centre e-mail: pv.followup@edaegypt.gov.eg or Egyptian Drug Authority (EDA) website: https://www.edaegypt.gov.eg or (EPVC) [hotline 15301].


Improving patient outcomes:
KEYTRUDA® for better cervical cancer survival
KEYTRUDA® offers new hope for improving survival in cervical cancer patients. Explore the latest updates on KN-A18 and how these can help you improve your practice.

See how we are growing hope in kidney cancer
At MSD, fighting kidney cancer is more than building a portfolio – it’s a commitment to therapeutic innovation, enhancing care, expanding possibilities and bringing hope to more patients.
References:
1- Egyptian Drug Authority KEYTRUDA leaflet approval date: 30/12/2024.
2- ESMO. ESMO-MCBS scorecards. 2024. Available at: https://www.esmo.org/guidelines/esmo-mcbs/esmo-mcbs-for-solid-tumours/esmo-mcbs-scorecards. Accessed July 27, 2025.
3- ESMO. ESMO-Magnitude of Clinical Benefit Scale factsheet. 2024. Available at: https://www.esmo.org/content/download/288505/5736229/1/ESMO-MCBS-Factsheet.pdf. Accessed July 27, 2025.
4- Garassino MC, Gadgeel S, Speranza G, et al. Pembrolizumab plus pemetrexed and platinum in nonsquamous non small-cell lung cancer: 5-year outcomes from the phase 3 KEYNOTE-189 study. J Clin Oncol. 2023; 41(11):1992-8.
5- Novello S, Kowalski DM, Luft A, et al. Pembrolizumab plus chemotherapy in squamous non small-cell lung cancer: 5-year update of the phase III KEYNOTE-407 study. J Clin Oncol. 2023; 41(11):1999-2006.
6- Reck M, Rodríguez-Abreu D, Robinson AG, et al. Five-year outcomes with pembrolizumab versus chemotherapy for metastatic non small-cell lung cancer with PD-L1 tumor proportion score ≥ 50. J Clin Oncol. 2021; 39(21):2339-49.
7- Barlesi F, Garon EB, Kim DW, et al. Health-related quality of life in KEYNOTE-010: a phase II/III study of pembrolizumab versus docetaxel in patients with previously treated advanced, programmed death ligand 1-expressing NSCLC. J Thorac Oncol. 2019; 14(5):793-801.
8- Brahmer JR, Rodríguez-Abreu D, Robinson AG, et al. Health-related quality-of-life results for pembrolizumab versus chemotherapy in advanced, PD-L1-positive NSCLC (KEYNOTE-024): a multicentre, international, randomised, open-label phase 3 trial. Lancet Oncol. 2017; 18(12):1600-9.
9- Garassino MC, Gadgeel S, Esteban E, et al. Patient-reported outcomes following pembrolizumab or placebo plus pemetrexed and platinum in patients with previously untreated, metastatic, non squamous non small-cell lung cancer (KEYNOTE-189): a multicentre, double-blind, randomised, placebo-controlled, phase 3 trial. Lancet Oncol. 2020; 21(3):387-97.
10- Pai-Scherf L, Blumenthal GM, Li H, et al. FDA approval summary: pembrolizumab for treatment of metastatic non small cell lung cancer: first-line therapy and beyond. Oncologist. 2017; 22(11):1392-9.
11- ClinicalTrials.gov. Study of pembrolizumab (MK-3475) in participants with progressive locally advanced or metastatic carcinoma, melanoma, or non-small cell lung carcinoma (KEYNOTE-001). 2011. Available at: https://clinicaltrials.gov/study/NCT01295827. Accessed July 27, 2025.
12- FDA. Pembrolizumab (KEYTRUDA) Checkpoint Inhibitor. 2016. Available at: https://www.fda.gov/drugs/resources-information-approved-drugs/pembrolizumab-keytruda-checkpoint-inhibitor. Accessed July 27, 2025.
13- Hendriks LE, Kerr KM, Menis J, et al. Non-oncogene-addicted metastatic non small-cell lung cancer: ESMO Clinical Practice Guideline for diagnosis, treatment and follow-up. Ann Oncol. 2023; 34(4):358-76.
14- Owen, D. H., Halmos, B., Puri, S., et al. Therapy for Stage IV Non-Small Cell Lung Cancer Without Driver Alterations: ASCO Living Guideline, Version 2025.1. Journal of clinical oncology: official journal of the American Society of Clinical Oncology, JCO2501062. Advance online publication. https://doi.org/10.1200/JCO-25-01062
15- National Comprehensive Cancer Network®. NCCN Clinical Practice Guidelines in Oncology (NCCN® Guidelines) for Non-Small Cell Lung Cancer V.8.2025. Accessed August 15, 2025.
16- Barlesi F, Garon EB, Kim DW, et al. Supplementary material. Health-related quality of life in KEYNOTE-010: a phase II/III study of pembrolizumab versus docetaxel in patients with previously treated advanced, programmed death ligand 1-expressing NSCLC. J Thorac Oncol. 2019; 14(5):793-801. Available at: https://www.jto.org/cms/10.1016/j.jtho.2019.01.016/attachment/68f75126-5ed6-4d4e-9d07-3224a9ec3c6a/mmc1.docx. Accessed July 27, 2025.
17- European Organisation for Research and Treatment of Cancer. FDA approves KEYTRUDA® (pembrolizumab) as adjuvant treatment following surgical resection and platinum-based chemotherapy for patients with stage IB (T2a ≥4 Centimeters), II, or IIIA non small cell lung cancer (NSCLC). 2023. Available at: https://www.eortc.org/blog/2023/01/30/fda-approves-keytruda-non-small-cell-lungcancer/. Accessed July 27, 2025.
18- Food & Drug Administration. FDA full Prescribing information for Keytruda, Available at: http://www.accessdata.fda.gov/drugsatfda_docs/label/2023/125514s139lbl.pdf. Accessed July 27, 2025.
19- European Medicines Agency. Keytruda – opinion on variation to marketing authorisation. Available at: https://www.ema.europa.eu/en/medicines/human/variation/keytruda-2. Accessed June 20, 2025.
20- ESMO website, KEYNOTE 407 Scorecard. Available at: https://www.esmo.org/guidelines/esmo-mcbs/esmo-mcbs-for-solid-tumours/esmo-mcbs-scorecards/scorecard-154-1. Accessed July 29, 2025
21- ESMO website, KEYNOTE 189 Scorecard. Available at: https://www.esmo.org/guidelines/esmo-mcbs/esmo-mcbs-for-solid-tumours/esmo-mcbs-scorecards/scorecard-132-1. Accessed July 29, 2025
22- ESMO website, KEYNOTE 024 Scorecard. Available at: https://www.esmo.org/guidelines/esmo-mcbs/esmo-mcbs-for-solid-tumours/esmo-mcbs-scorecards/scorecard-68-1. Accessed July 29, 2025
EG-KEY-00471 I Expiry date: 19/08/2026

